Inside my Sonicare toothbrush

Last week I took apart my old and recently-broken Sonicare toothbrush, to remove the batteries before throwing the rest away. I thought the insides were interesting, and that others might like to see.
I pried open the case with two flathead screwdrivers; it cracked cleanly down the sides. Then I snipped the wires connecting the circuit board, and popped out the batteries (which turned out to be really normal “AA” NiCad rechargeables, except that they had leads soldered directly to the contacts — This was actually quite a disappointment, since, with such standard batteries, they should be easily replaceable… but to get at them you have to tear the whole toothbrush apart, breaking the housing, and destroying the whole thing).


The inside contains:
- circuit board
- batteries
- two coils of wire, one on the bottom (next to where it would sit in the charger), and the other at the top (next to where the brush head attaches)
- a magnet at the base
… seems like the base is magnetically coupled to the charger, so that the current is passed from the charger to the coil in the toothbrush base via a magnetic field. This inductive coupling is not terribly efficient, but allows you to have a toothbrush that is completely sealed/waterproof, and that at least gives the perception of being safer due to a lack of metal contacts ((Draft Battery Charger Energy Star Standard)).
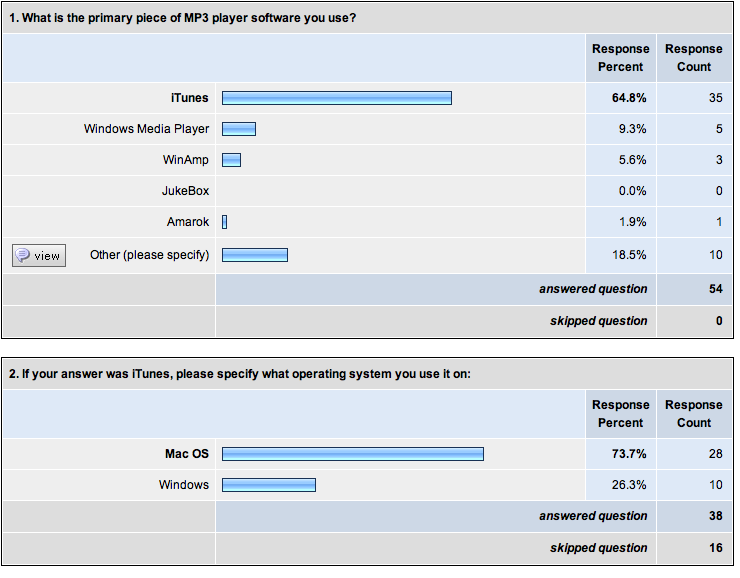
MP3 player software.
Here are the current results of the survey I posted last night:
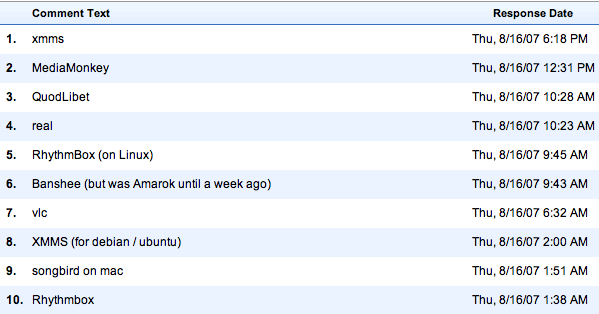
Here’s what the ten people in the “Other” category are using:
Thanks for all of your responses! Even though this is certainly not an unbiased sample set, it looks like building something for iTunes is a pretty safe way to go. Hooray for quantitative decision-making!
MP3 player software?
Maybe you guys can help me learn something… I’m wondering what MP3 player software my friends use.
I set up a very short (2-question) survey at SurveyMonkey:
What MP3 player software do you use?
It should only take 6-10 seconds. And the benefit will last a lifetime. Maybe.
Thanks so much to all who help by answering! Your input will help me decide what my next project — a visualization of your own music listening patterns — will focus on.
Addendum: One day later, I posted the survey results in this blog post.
Why older people die during heat waves
An article in yesterday’s New York Times, entitled “Sweatology”, talks about how important it is that we sweat. Sweat cools us off, yes. But the article also describes why different people sweat different amounts, and one factor is age.
“…beginning about age 60 both sexes sweat less, even if they are in good physical condition, and even if they become seriously overheated. Thus the statistics that during heat waves the elderly are at highest risk of heat stroke.”
I didn’t know I would sweat less when I get older. Maybe this means I should move to Florida now, and retire to San Francisco when I’m 60, instead of the other way around. And perhaps that I should spray my father intermittently with a misting bottle during his visit to Boston this week. Happy 60-plus-2-weeks, Dad!
I’m so lucky.
I’m trying to be better about reminding myself how lucky I am, in so many ways. I tend to forget these things, and I end up taking much of it for granted. One of the things I’ve certainly taken for granted is the place I went to college.
I’ve spent a few days during the past week working in the Stanford libraries — I’m out in the Bay area for a long vacation, and am trying to get some work done here and there.
It’s just amazingly beautiful here. The campus is huge, open, green, and sunny. Right now I’m on the second floor of Meyer, looking out at Hoover Tower and the enormous eucalyptus in front of Green Library. When I was here for school, I just thought this was normal… laying around on the grass, skating at night down the floors of the parking garages, cooking dinner for my co-op, jumping over to the city, sitting out in the sun next to the red fountain, dashing off on my bike from Mirrielees to the Med School. I was so absorbed in my personal life and classwork that I don’t know that I ever stopped to realize what I was a part of. It’s strange how you grow to take the most familiar things for granted.